Pharmacokinetics & ADME
Our comprehensive pharmacokinetic services provide detailed insights into how drugs interact with the body, from absorption to elimination.
Our team has extensive expertise in specialized surgical techniques that enable precise pharmacokinetic studies. These advanced procedures allow for targeted drug delivery and accurate sampling from specific anatomical sites.
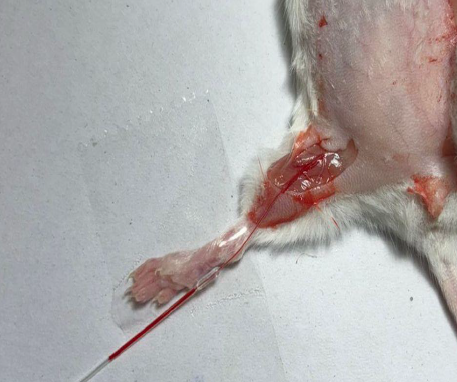
Femoral Artery Cannulation
Precise catheterization of the femoral artery allowing continuous blood sampling and monitoring of arterial drug concentrations with minimal disturbance to the animal.
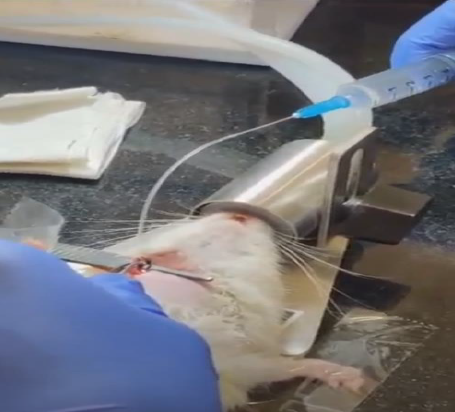
Jugular Vein Cannulation
Strategic placement of a catheter in the jugular vein enabling repeated blood sampling and intravenous drug administration for comprehensive PK profiling.
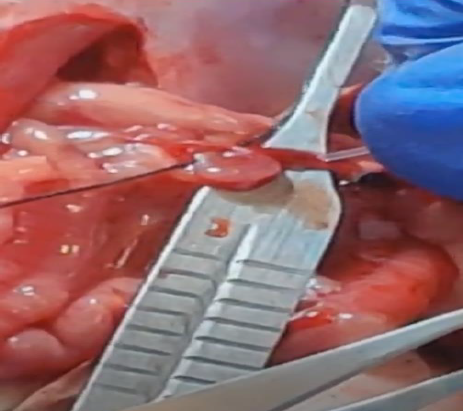
Triple Cannulation
Advanced technique involving simultaneous cannulation of the jugular vein, hepatic portal vein, and duodenum, allowing for comprehensive first-pass metabolism studies.
NEWAdvanced Surgical Capabilities
Our specialized surgical techniques enable precise pharmacokinetic studies that would otherwise be impossible. These procedures allow for:
- Direct sampling from specific anatomical sites
- Targeted drug delivery to organs of interest
- Continuous monitoring of drug concentrations
- Assessment of first-pass metabolism
- Evaluation of tissue-specific drug distribution
Our in vivo pharmacokinetic research provides comprehensive insights into drug behavior within living organisms, helping you make informed decisions throughout the drug development process.
Core PK Services
- Bioavailability assessment
- Tissue distribution studies
- PK/PD research correlation
- Dose escalation studies
- Dose accumulation studies
- Dose optimisation studies
Specialized Techniques
- PK on surgically cannulated rodents
- Jugular cannulation
- Femoral cannulation
- Bile duct cannulation
- Triple cannulation techniques
Administration Routes
Matrix Collection
We can collect and analyze various biological matrices including:
- Whole blood/Serum/Plasma
- Bone marrow
- Cerebrospinal fluid (limited sample volumes)
- Aqueous humor (limited sample volumes)
- Saliva sampling
- Portal blood Collection
- Blood sampling by cardiac puncture
Hepatic Models in Rat
Our specialized hepatic models provide comprehensive insights into liver disease progression and drug metabolism, enabling precise evaluation of hepatotoxicity and efficacy of liver-targeted therapeutics.
Liver Disease Progression Models
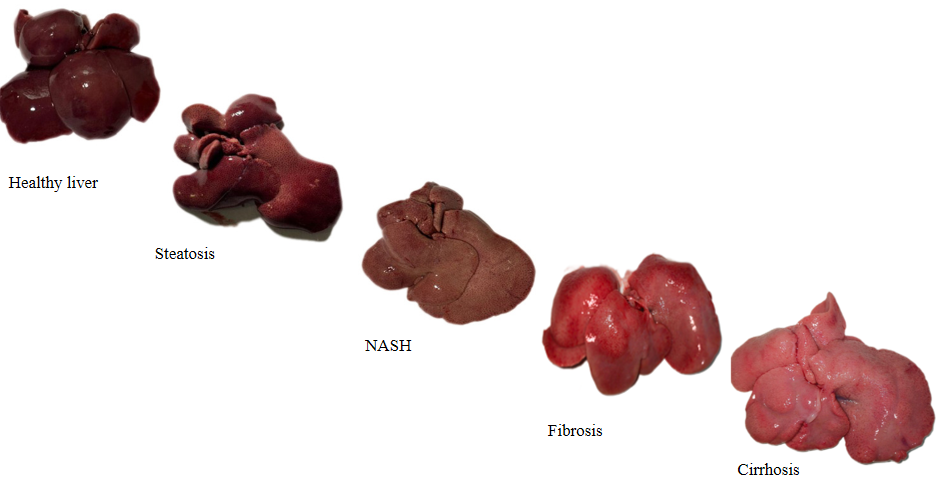
Healthy Liver
Baseline control
Steatosis
Fatty liver
NASH
Non-alcoholic steatohepatitis
Fibrosis
Scarring stage
Cirrhosis
Advanced scarring
Applications:
- Evaluation of hepatoprotective compounds
- Assessment of drug-induced liver injury (DILI)
- Pharmacokinetic studies in compromised liver function
- Development of therapeutics for NAFLD/NASH
- Investigation of liver regeneration mechanisms
Our specialized hepatic models provide comprehensive insights into liver disease progression and drug metabolism, enabling precise evaluation of hepatotoxicity and efficacy of liver-targeted therapeutics.
Liver Disease Progression Models

Healthy Liver
Baseline control
Steatosis
Fatty liver
NASH
Non-alcoholic steatohepatitis
Fibrosis
Scarring stage
Cirrhosis
Advanced scarring
Applications:
- Evaluation of hepatoprotective compounds
- Assessment of drug-induced liver injury (DILI)
- Pharmacokinetic studies in compromised liver function
- Development of therapeutics for NAFLD/NASH
- Investigation of liver regeneration mechanisms
Pharmacokinetic Studies for Finished Dosage Forms
We successfully designed and executed PK studies for sublingual films, sublingual tablets, nasal sprays, and eye drops to support clients' 505(b)(2) applications, patent filings, and clinical trial entry.
Key Achievements:
- Developed customized PK protocols to assess absorption, bioavailability, and systemic exposure
- Provided rapid and precise data analysis to meet regulatory expectations
- Enabled clients to achieve key milestones, including patent filings and advancing select candidates into clinical trials
- Ensured cost-effective and regulatory-compliant study designs to accelerate drug development
Comprehensive Analysis
Full ADME profiling
Multiple Administration Routes
Specialized delivery methods
Regulatory Compliance
Supporting documentation for submissions